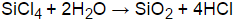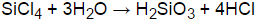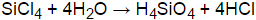SiCl4 + (2+n)H2O → SiO2•nH2O + 4HCl
Silicon chloride react with water to produce silicic acids and hydrogen chloride.
Individual reactions:
Elementary acts:
Individual reactions:
Elementary acts:
- a) SiCl4 + H2O → [Si(H2O)Cl4] (activated complex)
- [Si(H2O)Cl4] → SiCl3(OH) + HCl
- b) SiCl3(OH) + H2O → [Si(H2O)Cl3(OH)] (activated complex)
- [Si(H2O)Cl3(OH)] → SiCl2(OH)2 + HCl
- c) SiCl2(OH)2 → SiO2 + 2HCl
- d) 2SiCl3(OH) → Cl3Si-O-SiCl3 + H2O
- Cl3Si-O-SiCl3 + 3H2O → 2SiO2 + 6HCl